Nexera Mikros - Vlastnosti
Supporting Micro Flowrate Range Liquid Chromatography Mass Spectrometry System
Improved Detection Limits with Microflow
High Bioanalytical Sensitivity
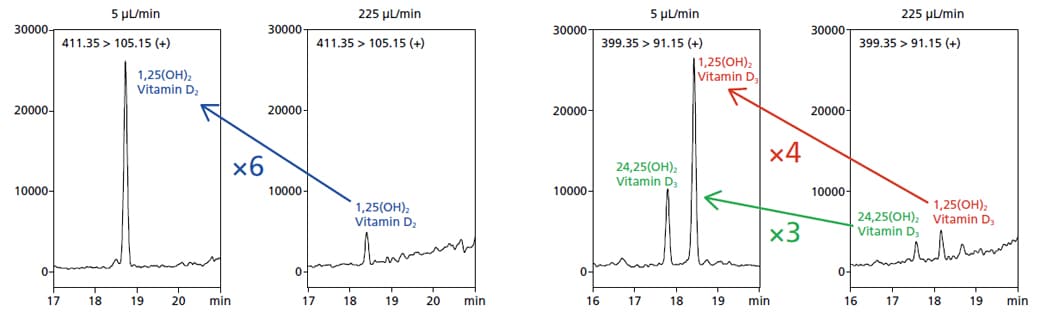
Sensitivity remains a challenge for many vitamin D metabolites, such as 1,25(OH)2 di-hydroxyvitamin D and 24,25(OH)2 di-hydroxyvitamin D, because they lack easily ionizable functional groups and are in low-abundance. As shown below, microflow LC/MS/MS analysis of standard solutions of the aforementioned vitamin D analytes resulted in a three- to five-fold improvement in the signal-to-noise ratio when compared to semi-microflow analysis.
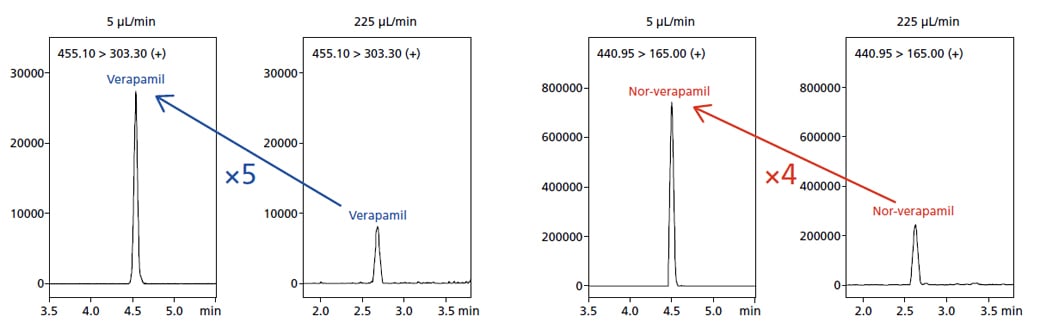
The increased sensitivity achievable by microflow LC/MS/MS, compared to semi-microflow, resulted in a five-fold increase in the signal-to-noise ratio for Verapamil and a four-fold increase in the signal-to-noise ratio of Nor-Verapamil.
Monoclonal Antibody Bioanalysis
- Nexera™ Mikros and nSMOL™ Antibody BA Kit -
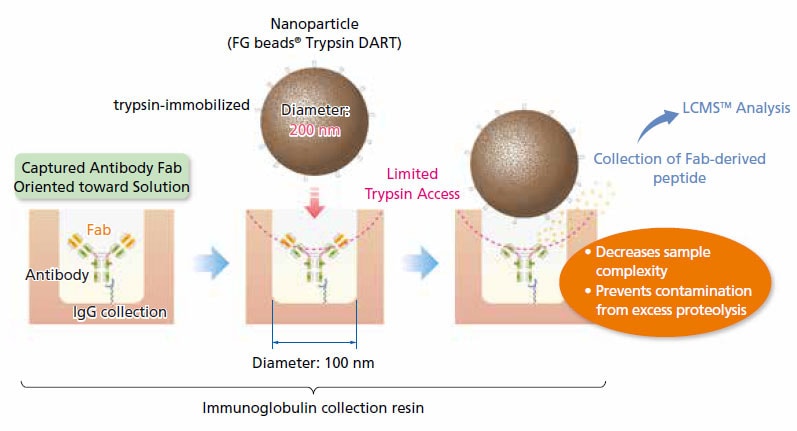
nSMOL (nano-surface and molecular orientation limited proteolysis) is a proprietary, ground-breaking process that enables selective proteolysis of the Fab region of antibodies using trypsin-immobilized nanoparticles. Fab-derived peptides are quantified via MRM measurement on a high-sensitivity mass spectrometer. The Nexera Mikros system achieved signal intensities 12 times higher than with conventional flow rates, with a corresponding lower limit of detection of 0.025 μg/mL and good linearity. The Nexera Mikros system is ideal for low-level quantitation of peptides by LCMS.
The following is an example of antibody drug analysis in plasma. The plasma spiked with trastuzumab was pretreated using nSMOL Antibody BA Kit.
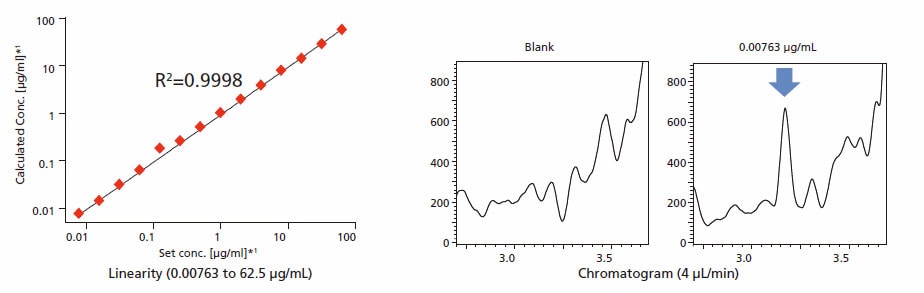
In the analysis of signature peptides deriving from trastuzumab, Nexera Mikros provides a dynamic range of 0.00763 - 62.5 μg/mL, good linearity with R2 > 0.99, and excellent accuracy of 101.0% (average).
The intra-day reproducibility of this trastuzumab-derived-peptide analysis is summarized in the table below.
These results show an excellent reproducibility, with both of accuracy and precision within 20 % at LLOQ and within 15 % even at other concentrations.
| Concentration Setting (µg/mL) |
QC set1 *2 | QC set2*2 | ||
|---|---|---|---|---|
| Accuracy | Repeatability | Accuracy | Repeatability | |
| 0.00763 | 97.1% | 5.69% | 100% | 11.3% |
| 0.0229 | 102% | 6.68% | 101% | 2.84% |
| 5.86 | 106% | 2.67% | 99.4% | 3.12% |
| 50.0 | 94% | 6.36% | 91.7% | 7.23% |
Intra-Day Reproducibility Evaluation Results from QC Samples
*1: This curve is drawn on logarithmic scale.
*2: QC sets 1 and 2 were analyzed over a 2-day period, with each set of concentrations analyzed five times.
Incomparable Microflow Ruggedness
Realize the Benefits of Microflow While Enjoying the Ruggedness and Reliability of HPLC
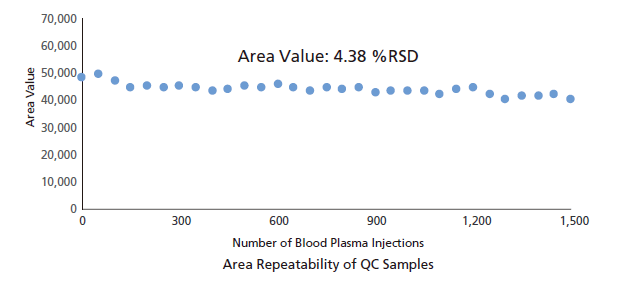
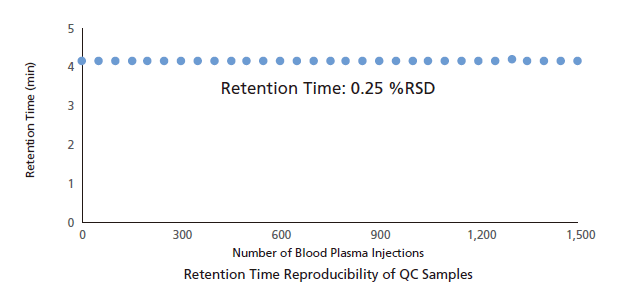
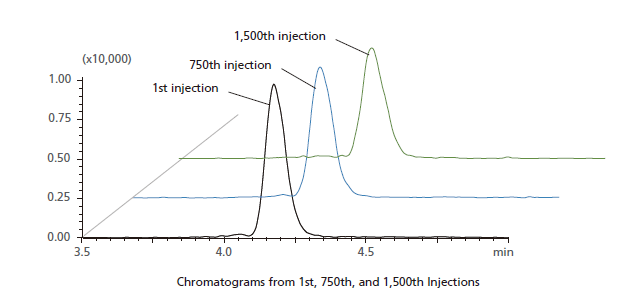
The Nexera Mikros system delivers the improvements you expect from a low-flow system with the ruggedness of HPLC. A plasma blank was protein precipitated, diluted (3x), and injected 1,500 times consecutively. During the consecutive injections, QC sample (40 ppt of nortriptyline) was injected once every 50 injections of plasma blank. Peak areas and retention times of QC sample are plotted below. With 4.38% RSD area repeatability and 0.25% RSD retention time stability, extremely stable operation was demonstrated, without loss of peak shape common in high-throughput analyses of complex matrices.
*Results obtained under our application conditions.
Easy, Familiar Operation
Robust and Easy-to-Use Microflow ESI Source
Micro-ESI™ Sources
Shimadzu’s Micro-ESI LCMS source has been designed for optimal sensitivity and ease of use. An X–Y adjustable stage and viewing camera allow for easy ESI spray needle adjustment. The column oven is mounted on the LCMS to minimize the volume between the exit of the column and the ESI needle.
The Micro-ESI probe also features an ESI spray angle optimized for microflow rates, and is available for triple quadrupole mass detection mass spectrometryand quadrupole time-of-flight mass spectrometry, respectively.

CTO-Mikros™ + UF-Link™: Easy, Dependable Column Installation
Mounted to the Micro-ESI source is the CTO-Mikros column oven. Stable column temperature, essential to retention time reproducibility, is ensured and columns up to 150 mm in length can be accommodated. To preserve the optimal source adjustment, the CTO-Mikros features an innovative snap-in column locking system, the UF-Link.
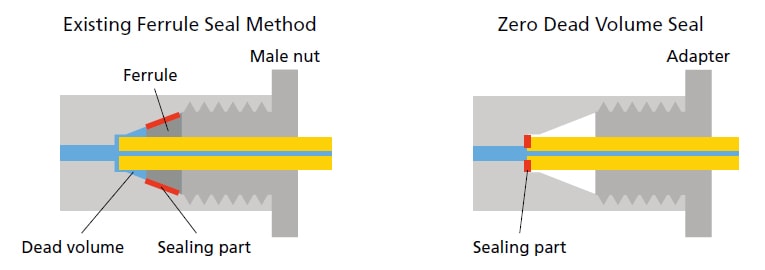
When using microfow rates, even the smallest of dead volumes can cause significant diffusion and lead to loss of peak shape and sensitivity. Shimadzu’s UF-Link allows any user to correctly install a microflow column without disrupting the ESI spray needle position. A sure-sealing adapter allows the column to lock into the oven and make a zero-dead volume connection with the ESI needle. Any column up to 150 mm in length with standard fittings can be accommodated.
Connection Procedure

Attach the adapter to the column.
Standard threads on the adapter make
it compatible with a wide variety of
columns.

Place the column in the UF-Link
mechanism inside the oven.

Complete the connection by swinging
over the lever.
LC-Mikros™: Innovative Microflow Pump Design

Shimadzu LC pumps are the industry standard for long life and high performance. The LC-Mikros solvent delivery pump has completely new direct-drive engineering to deliver micro to semi-micro flow rates ranging from 1 to 500 μL/min. UHPLC system pressure tolerance of 80 MPa (11,600 psi) allows the use of every column type.
The LC-Mikros pump is designed for microflow gradient elution, even at low percentages. In the example below, excellent gradient stability is shown using mobile phase compositions ranging from 1% to 90%. The LC-Mikros delivers stable retention times essential to the chromatographer.
-
Average
Retention TimeStandard
DeviationRSD Gly-Tyr 5.31 0.009 0.16% Val-Tyr-Val 7.97 0.006 0.07% Angiotensin II 9.32 0.015 0.16% Leu enkephalin 9.71 0.011 0.11% Met enkephalin 10.5 0.012 0.11% -
Flowrate: 5 µL/min
Column: Shim-pack MC C18 (0.3 mmID×150 mm)
*Obtained under our specified conditions.
| Time (min) | 1 | 11 | 11.1 | 13 | 13.1 | 25 |
|---|---|---|---|---|---|---|
| Concentration B | 1% | 40% | 90% | 90% | 1% | 1% |
SIL-40C XR: Low-Volume Direct Injection Autosampler

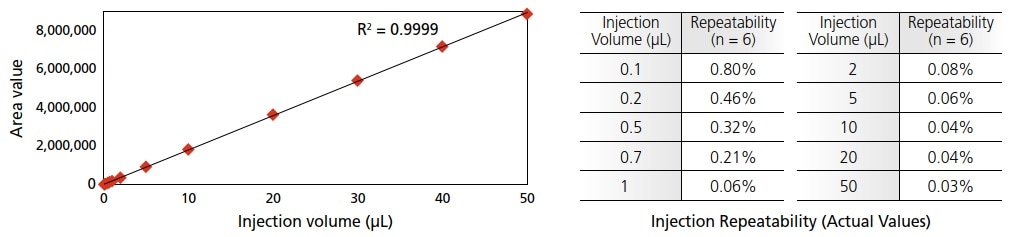
The SIL-40C XR autosampler delivers you excellent accuracy and reproducibility as well as extremely low carry-over. Its injection volume ranges from 0.1 to 50 μL and your precious sample has never wasted due to the total injection method SIL-40C XR adapted.
It contributes to produce highly accurate results even if you had to use low injection volume (less than 1 μL). In case of micro flow rate with narrow column, optimum injection volume also gets lowered. It is also difficult to increase the injection volume, especially, if you would use a sample solvent with high elution power. In this case, sample must be diluted to weaken the sample solvents. Highly accurate micro volume injection capability of SIL-40C XR allows you to use samples with high organic solvent contents without dilution.
System volume and gradient delay are minimized with the newly developed “programmable loop cut-off mechanism,” which automatically diverts the sample loop from the flow line using a high-pressure valve as soon as the sample in needle is drained. This is extremely important in minimizing microflow analysis run times.